Genentech Says FDA Gives Accelerated Approval to Tecentriq Plus Chemotherapy
March 08 2019 - 1:10PM
Dow Jones News
By Stephen Nakrosis
Genentech on Friday said the U.S. Food and Drug Administration
granted accelerated approval to Tecentriq plus chemotherapy for
certain adult breast cancer patients.
The approval was based on data from the Phase III IMpassion130
study, which demonstrated that Tecentriq plus certain chemotherapy
drugs significantly reduced the risk of the disease worsening or
death, the company said. Safety appeared consistent with the known
safety profiles of the individual medicines, and no new safety
signals were identified with the combination, the company said.
Genentech said this Tecentriq combination is the first cancer
immunotherapy regimen approved for breast cancer.
The FDA's Accelerated Approval Program can give faster approval
of treatments for serious conditions where there is an unmet
medical need.
Genentech is a member of the Roche Group.
Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
March 08, 2019 12:55 ET (17:55 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
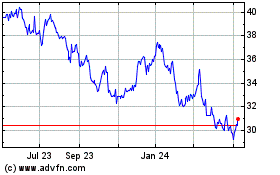
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From Mar 2024 to Apr 2024
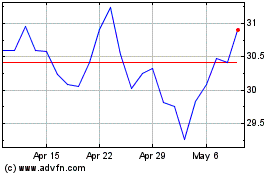
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From Apr 2023 to Apr 2024