Study expected to commence in first half of
2020
AVITA Medical Limited (ASX: AVH, NASDAQ: RCEL), a regenerative
medicine company with a technology platform positioned to address
unmet medical needs in therapeutic skin restoration, announced
today that the U.S. Food and Drug Administration (FDA) has approved
the company’s Investigational Device Exemption (IDE) application to
conduct a feasibility study evaluating the safety and effectiveness
of the RECELL® Autologous Cell Harvesting Device (RECELL® System)
for repigmentation of depigmented lesions associated with stable
vitiligo.
“Vitiligo affects approximately 6.5 million people in the United
States(i), rivalling the prevalence of psoriasis(ii); however,
there are limited treatment options available to patients to
permanently restore skin pigmentation,” said Dr. Mike Perry, AVITA
Medical Chief Executive Officer. “We’re pleased with the FDA’s
decision which allows us to begin our vitiligo study in the first
half of 2020. Based on the outcomes of the feasibility study, we
anticipate proceeding with a pivotal clinical trial to pursue FDA
approval of the RECELL System as a cell-based repigmentation
treatment option for stable vitiligo.”
Vitiligo is a disease resulting in loss of color, or
pigmentation, in patches of skin that impacts the quality of life
for those living with the condition.(iii) There is currently no
cure for vitiligo, nor a universally accepted method for limiting
the spread of the disease. Although many treatments are being used
for the management of vitiligo, they are often temporary with a
high rate of recurrence.(iv)
“This study expands on peer-reviewed, published effectiveness
outcomes to confirm the feasibility of RECELL as a treatment for
repigmentation in cases of stable vitiligo,” said Andy Quick, AVITA
Medical’s Chief Technology Officer. “Given the RECELL System’s
broad approval outside of the U.S., more than 1,000 vitiligo
patients have already been treated globally and reported
repigmentation.”
AVITA Medical will collaborate with a leading medical center to
conduct a pilot study with 10 patients who have vitiligo lesions
that have been stable for at least one year. Areas of the vitiligo
lesion will be randomly treated with slightly varying cell
suspensions prepared using RECELL to confirm response rates and
optimal suspension parameters.
The randomized controlled study’s primary effectiveness measure
is the percent area of repigmented skin 24 weeks after treatment,
as evaluated by a clinician blinded to the treatment assignment.
Additional effectiveness data collected over the course of the
24-week study will include degree of repigmentation achieved and
patient rating of repigmentation.
Of note: Use of the RECELL System in patients undergoing
reconstruction of skin defects not associated with a burn injury is
limited by Federal law to investigational use.
ABOUT AVITA MEDICAL LIMITED
AVITA Medical is a regenerative medicine company with a
technology platform positioned to address unmet medical needs in
burns, chronic wounds, and aesthetics indications. AVITA Medical’s
patented and proprietary collection and application technology
provides innovative treatment solutions derived from the
regenerative properties of a patient’s own skin. The medical
devices work by preparing a REGENERATIVE EPIDERMAL SUSPENSION™
(RES™), an autologous suspension comprised of the patient’s skin
cells necessary to regenerate natural healthy epidermis. This
autologous suspension is then sprayed onto the areas of the patient
requiring treatment.
AVITA Medical’s first U.S. product, the RECELL® System, was
approved by the U.S. Food and Drug Administration (FDA) in
September 2018. The RECELL System is indicated for use in the
treatment of acute thermal burns in patients 18 years and older.
The RECELL System is used to prepare Spray-On Skin™ Cells using a
small amount of a patient’s own skin, providing a new way to treat
severe burns, while significantly reducing the amount of donor skin
required. The RECELL System is designed to be used at the point of
care alone or in combination with autografts depending on the depth
of the burn injury. Compelling data from randomized, controlled
clinical trials conducted at major U.S. Burn Centers and real-world
use in more than 8,000 patients globally, reinforce that the RECELL
System is a significant advancement over the current standard of
care for burn patients and offers benefits in clinical outcomes and
cost savings. Healthcare professionals should read the INSTRUCTIONS
FOR USE - RECELL® Autologous Cell Harvesting Device
(https://recellsystem.com/) for a full description of indications
for use and important safety information including
contraindications, warnings and precautions.
In international markets, our products are marketed under the
RECELL System brand to promote skin healing in a wide range of
applications including burns, chronic wounds and aesthetics. The
RECELL System is TGA-registered in Australia and received CE-mark
approval in Europe. To learn more, visit www.avitamedical.com
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This letter includes forward-looking statements. These
forward-looking statements generally can be identified by the use
of words such as “anticipate,” “expect,” “intend,” “could,” “may,”
“will,” “believe,” “estimate,” “look forward,” “forecast,” “goal,”
“target,” “project,” “continue,” “outlook,” “guidance,” “future,”
other words of similar meaning and the use of future dates.
Forward-looking statements in this letter include, but are not
limited to, statements concerning, among other things, our ongoing
clinical trials and product development activities, regulatory
approval of our products, the potential for future growth in our
business, and our ability to achieve our key strategic, operational
and financial goal. Forward-looking statements by their nature
address matters that are, to different degrees, uncertain. Each
forward- looking statement contained in this letter is subject to
risks and uncertainties that could cause actual results to differ
materially from those expressed or implied by such statement.
Applicable risks and uncertainties include, among others, the
timing of regulatory approvals of our products; physician
acceptance, endorsement, and use of our products; failure to
achieve the anticipated benefits from approval of our products; the
effect of regulatory actions; product liability claims; risks
associated with international operations and expansion; and other
business effects, including the effects of industry, economic or
political conditions outside of the company’s control. Investors
should not place considerable reliance on the forward-looking
statements contained in this letter. Investors are encouraged to
read our publicly available filings for a discussion of these and
other risks and uncertainties. The forward-looking statements in
this letter speak only as of the date of this release, and we
undertake no obligation to update or revise any of these
statements. This press release was authorized by the review
committee of AVITA Medical.
_________________________ (i) Advances in Vitiligo: An Update on
Medical and Surgical Treatments. A. Dillon, et al. J Clin Aesth
Derm. 2017 (ii) National Psoriasis Foundation – Statistics,
https://www.psoriasis.org/content/statistics Accessed 12/28/19
(iii) Willingness‐to‐pay and quality of life in patients with
vitiligo. Radtke, et al. BJD. 2009 (iv) Vitiligo Research
Foundation – Treatment Guidelines.
https://vrfoundation.org/treatment_guidelines Accessed 12/28/19
View source
version on businesswire.com: https://www.businesswire.com/news/home/20191229005010/en/
U.S. Media Sam Brown, Inc. Christy Curran Phone
+1-615-414-8668 christycurran@sambrown.com
O.U.S. Media Rudi Michelson Phone +61 (0)3 9620 3333
Mobile +61 (0)411 402 737 rudim@monsoon.com.au
Investors Westwicke Partners Caroline Corner Phone
+1-415-202-5678 caroline.corner@westwicke.com
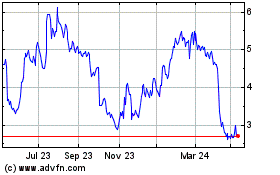
AVITA Medical (ASX:AVH)
Historical Stock Chart
From Mar 2024 to Apr 2024
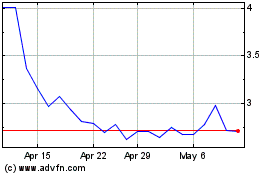
AVITA Medical (ASX:AVH)
Historical Stock Chart
From Apr 2023 to Apr 2024